Hypovolemic POTS: Symptoms, Causes, and Treatment for Low Blood Volume POTS

Is Your POTS Driven by Low Blood Volume?
Up to 70% of POTS patients have measurable hypovolemia. Use this interactive guide to understand the subtypes, check your risk factors, and learn what to do next.
Hypovolemic POTS
Found in ~45-70% of patientsNeuropathic POTS
~50% show sudomotor abnormalitiesHyperadrenergic POTS
Standing norepinephrine ≥600 pg/mLSymptom Prevalence in POTS Patients
Cognitive FX's 5-day POTS/ANS Therapy Program identifies which neurological systems are misfiring and targets them directly, addressing the root cause rather than just the symptoms.
Schedule a Consultation- Raj SR, Biaggioni I, Yamhure PC, et al. Renin-aldosterone paradox and perturbed blood volume regulation underlying postural tachycardia syndrome. Circulation. 2005;111(13):1574-1582. PMID: 15781744
- Fu Q, Vangundy TB, Galbreath MM, et al. Cardiac origins of the postural orthostatic tachycardia syndrome. J Am Coll Cardiol. 2010;55(25):2858-2868. PMID: 20579544
- Sheldon RS, Grubb BP, Olshansky B, et al. 2015 Heart Rhythm Society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome. Heart Rhythm. 2015;12(6):e41-e63. PMID: 25980576
- Vernino S, Bourne KM, Stiles LE, et al. Postural orthostatic tachycardia syndrome (POTS): State of the science and clinical care from a 2019 National Institutes of Health Expert Consensus Meeting - Part 1. Auton Neurosci. 2021;235:102828. PMID: 34144933
- Angeli AM, Salonen BR, Ganesh R, et al. Symptom presentation by phenotype of postural orthostatic tachycardia syndrome. Sci Rep. 2024;14(1):205. PMID: 38168762
- Raj SR, Black BK, Biaggioni I, et al. Propranolol decreases tachycardia and improves symptoms in the postural tachycardia syndrome. Circulation. 2009;120(9):725-734. PMID: 19687359
- Fu Q, Levine BD. Exercise and non-pharmacological treatment of POTS. Auton Neurosci. 2018;215:20-27. PMID: 30001836
- Wells R, Malik V, Brooks AG, et al. Cerebral blood flow and cognitive performance in postural tachycardia syndrome: insights from sustained cognitive stress test. J Am Heart Assoc. 2020;9(24):e017861. PMID: 33280488
- Shaw BH, Stiles LE, Bourne K, et al. The face of postural tachycardia syndrome - insights from a large cross-sectional online community-based survey. J Intern Med. 2019;286(4):438-448. PMID: 30861229
- Coffin ST, Black BK, Biaggioni I, et al. Desmopressin acutely decreases tachycardia and improves symptoms in the postural tachycardia syndrome. Heart Rhythm. 2012;9(9):1484-1490. PMID: 22561596
- Taub PR, Zadourian A, Lo HC, et al. Randomized trial of ivabradine in patients with hyperadrenergic postural orthostatic tachycardia syndrome. J Am Coll Cardiol. 2021;77(7):861-871. PMID: 33602468
- Ormiston CK, Swiatkiewicz I, Taub PR. Postural orthostatic tachycardia syndrome as a sequela of COVID-19. Heart Rhythm. 2022;19(11):1880-1889. PMID: 35853576
- Wang E, Ganti T, Vaou E, Hohler A. The relationship between mast cell activation syndrome, postural tachycardia syndrome, and Ehlers-Danlos syndrome. Allergy Asthma Proc. 2021;42(3):243-246. PMID: 33980338
- Fedorowski A. Postural orthostatic tachycardia syndrome: clinical presentation, aetiology and management. J Intern Med. 2019;285(4):352-366. PMID: 30372565
- Boris JR, Shadiack EC, McCormick EM, et al. Long-term POTS outcomes survey: diagnosis, therapy, and clinical outcomes. J Am Heart Assoc. 2024;13(14):e033485. PMID: 38958137
- Nwazue VC, Raj SR. Confounders of vasovagal syncope: postural tachycardia syndrome. Cardiol Clin. 2013;31(1):89-100. PMID: 23217690
Up to 70% of people with postural orthostatic tachycardia syndrome (POTS) are walking around with measurably less blood than they should have. The average deficit is about 13%, or roughly 700 mL — nearly a pint and a half missing from the circulatory system. That blood volume shortfall is the defining feature of hypovolemic POTS, and it drives everything from the racing heart on standing to the brain fog that keeps patients from functioning. Despite being the most common mechanism behind POTS, hypovolemia is still under-tested and under-treated by most clinicians who encounter this condition.
This guide breaks down what hypovolemic POTS actually is, why it happens, how it feels, and what can be done about it.
What is hypovolemic POTS?
POTS is diagnosed when heart rate jumps 30 or more beats per minute within 10 minutes of standing (40 bpm in adolescents), without a significant drop in blood pressure, and symptoms have persisted for at least three months. The 2015 Heart Rhythm Society Expert Consensus Statement formalized these criteria, and the 2020 Canadian Cardiovascular Society Position Statement added important refinements: the baseline supine heart rate must be at least 60 bpm, and the tachycardia must be sustained across multiple measurements rather than a single spike.
Within POTS, three overlapping pathophysiological mechanisms have been identified: neuropathic (damaged small nerve fibers that fail to constrict blood vessels), hyperadrenergic (excessive norepinephrine release creating sympathetic overdrive), and hypovolemic (reduced circulating blood volume). These are not mutually exclusive categories. A 2024 prospective study of 378 patients at a subspecialty POTS clinic found that 41.7% had two mechanisms operating simultaneously and 11.4% had all three.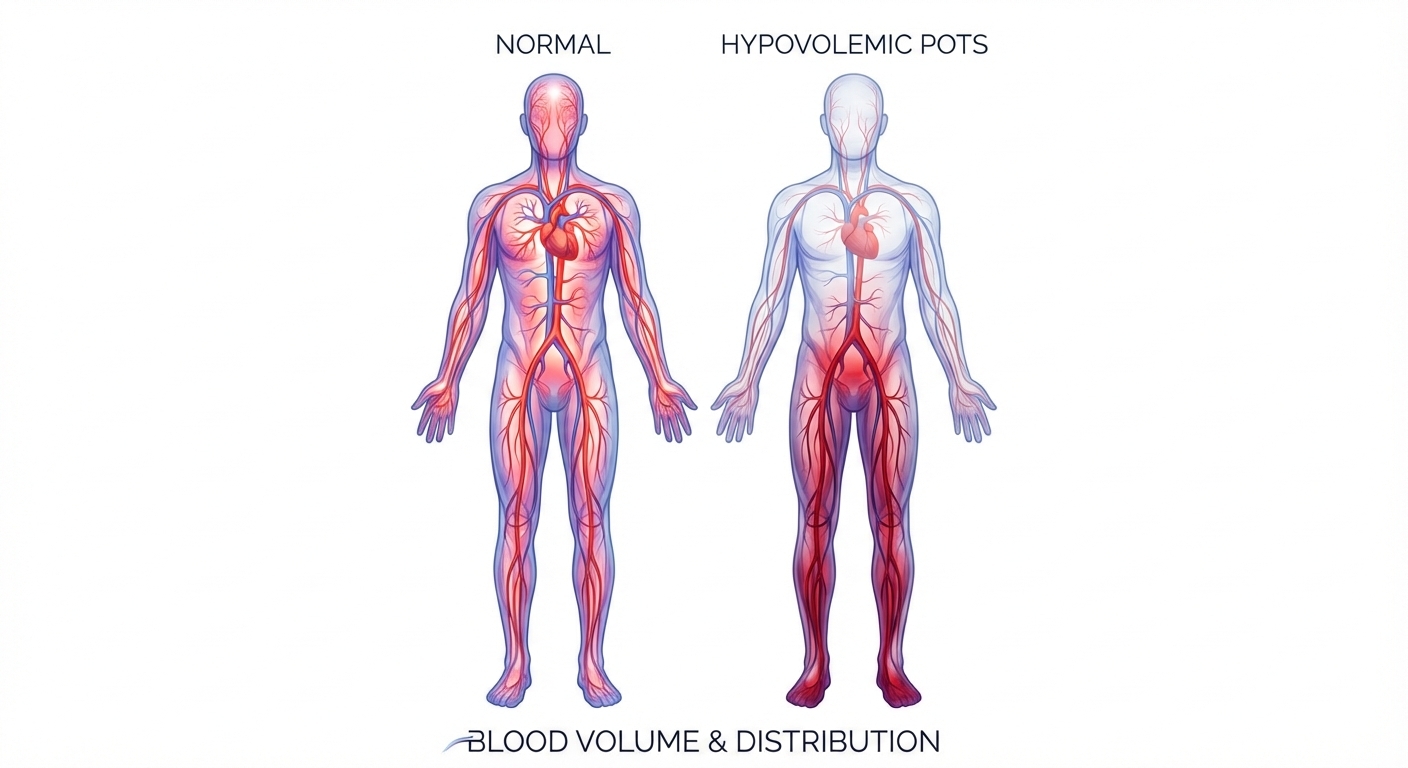
Hypovolemic POTS is not a separately coded diagnosis. It is a phenotypic descriptor that identifies which mechanism is driving a patient's symptoms. Johns Hopkins Medicine defines it simply as POTS associated with abnormally low blood volume. Identifying it matters because it changes the treatment approach: patients with a volume deficit need volume-targeted interventions, not just rate control or vasoconstriction.
In the 378-patient study, hypovolemic POTS was present in 44.9% of those tested, identified by a 24-hour urine sodium below 100 mmol — a practical marker of inadequate salt and water retention by the kidneys.
Why does blood volume drop in hypovolemic POTS?
The short answer is that the kidneys fail to hold onto salt and water even when the body desperately needs them to. The long answer involves a paradox that Vanderbilt University researchers identified in a landmark 2005 Circulation study, and it remains one of the most important findings in POTS pathophysiology.
In healthy people, low blood volume triggers a hormonal cascade called the renin-angiotensin-aldosterone system (RAAS). The kidneys release renin, which converts to angiotensin, which stimulates the adrenal glands to release aldosterone, which tells the kidneys to reabsorb sodium and water. Blood volume goes up. Problem solved.
In hypovolemic POTS, this system breaks. Satish Raj's Vanderbilt team measured blood volume deficits averaging 689 mL in POTS patients compared to controls, yet their plasma renin activity was identical to healthy subjects and their aldosterone was cut in half (190 vs. 380 pmol/L). The kidneys were dumping salt and water as if the patient were overhydrated, when the opposite was true.
Later work found that angiotensin II was actually elevated in POTS patients while the enzyme that breaks it down (ACE2) was suppressed. The signal to retain volume was present but garbled — high angiotensin II somehow failed to trigger adequate aldosterone release. This "renin-aldosterone paradox" is now considered the core neurohumoral defect in hypovolemic POTS.
Both components of blood are reduced. The same Vanderbilt study documented plasma volume deficits of 334 mL and red blood cell volume deficits of 356 mL. A 2024 Vanderbilt validation study using CO-rebreathing (a non-invasive measurement method) confirmed total blood volume deviation of about -14% from predicted normal, with a strong correlation between volume deficit and standing heart rate: the more blood missing, the faster the heart races.
Several factors compound the volume deficit:
Venous pooling. When a hypovolemic POTS patient stands, blood shifts rapidly into the legs, pelvis, and splanchnic (abdominal) circulation. With less total volume to start, the heart sees an even steeper drop in returning blood. Stroke volume falls. Heart rate compensates by surging upward.
Cardiac deconditioning. Chronic low blood volume leads to reduced cardiac filling, which over time causes the heart to shrink. Researchers at UT Southwestern found left ventricular mass about 16% below normal in POTS patients, with peak oxygen consumption (VO2) of just 26 mL/kg/min versus 36 in controls. A smaller heart moves less blood per beat, requiring still more tachycardia.
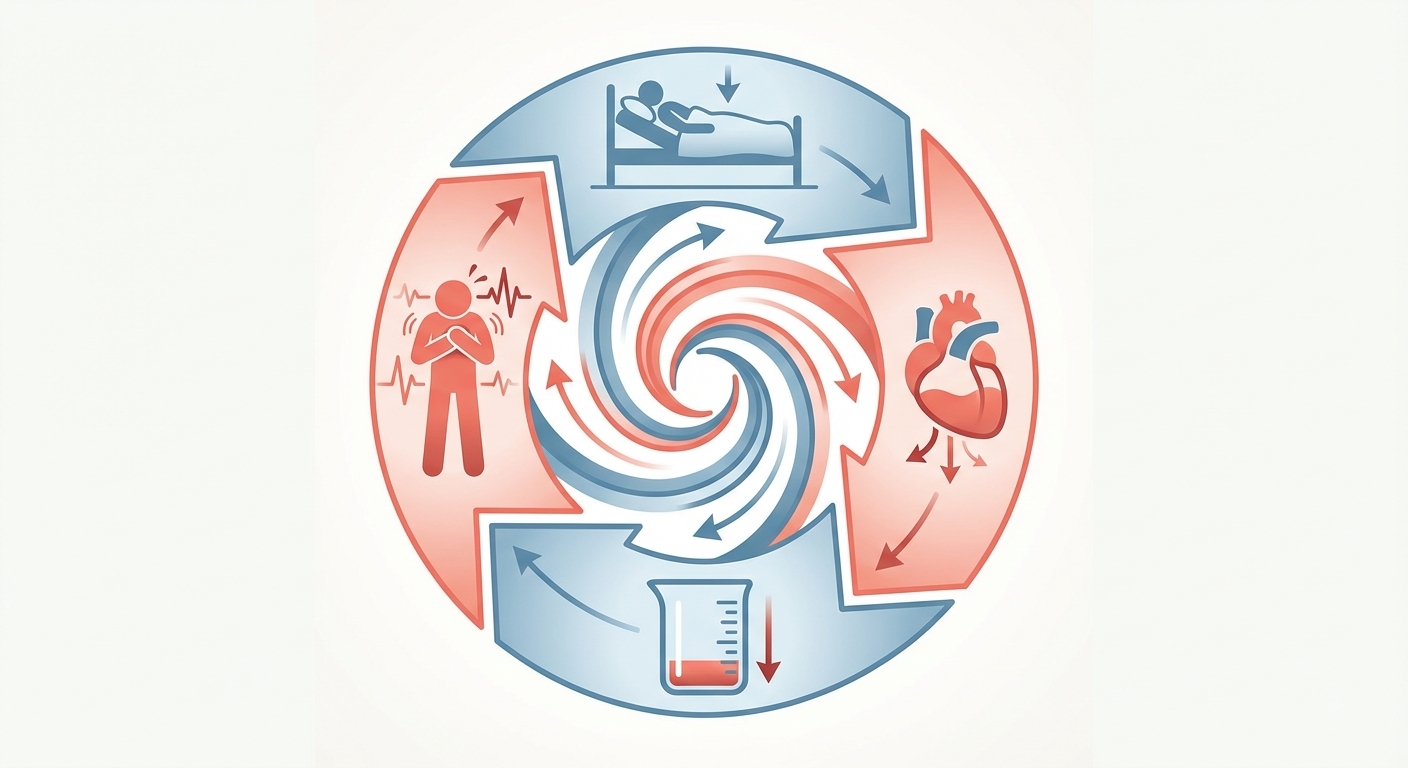
The deconditioning spiral. Symptoms discourage activity. Inactivity worsens deconditioning. Deconditioning reduces blood volume further. The spiral feeds itself, and it can progress rapidly — weeks of bedrest can reduce blood volume by 10-15%.
What does hypovolemic POTS feel like?
The symptoms of hypovolemic POTS extend far beyond a fast heartbeat. In the 378-patient phenotyping study, lightheadedness was reported by 97.6% of patients and rated the most disabling symptom by nearly a third. Patients lost an average of 3.0 days per week to symptoms and experienced 4.7 unproductive days — disability comparable to congestive heart failure.
Orthostatic symptoms are the most recognizable: heart racing, pounding, or fluttering when standing; dizziness that worsens in the first few minutes upright; visual graying or tunneling; presyncope (the sensation of nearly fainting); and, in some patients, actual syncope. Heat, prolonged standing, large meals, and morning hours typically worsen these symptoms.
Brain fog is often the most functionally devastating symptom. It is not a vague complaint — it is a measurable consequence of cerebral hypoperfusion. A 2020 Journal of the American Heart Association study showed cerebral blood flow velocity dropped 7.8% in POTS patients during sustained cognitive tasks (versus 1.8% in controls), with a corresponding 6.1% slowing in psychomotor speed. Critically, this happened without any positional change — cognitive effort alone was enough to deplete already-marginal cerebral perfusion. Patients describe difficulty concentrating, word-finding problems, slowed processing, and feeling mentally exhausted after routine cognitive tasks.
Fatigue in hypovolemic POTS is not ordinary tiredness. It is a bone-deep exhaustion that does not resolve with sleep and often worsens with physical or cognitive exertion. Many patients meet criteria for chronic fatigue syndrome simultaneously.
Gastrointestinal symptoms are common and frequently underappreciated. Nausea, bloating, early satiety, abdominal pain, and irregular bowel patterns arise from reduced splanchnic blood flow, autonomic disruption of gut motility, and in patients with concurrent mast cell activation, mediator-driven GI inflammation. Gastroparesis (delayed stomach emptying) overlaps in a significant subset.
Exercise intolerance is both a symptom and a perpetuating factor. Patients report rapid heart rate escalation with minimal effort, prolonged recovery after activity, and in some cases post-exertional worsening of all symptoms lasting hours to days.
Other autonomic symptoms include temperature dysregulation (heat intolerance, cold extremities), disrupted sleep architecture, coat-hanger pain (aching in the neck and shoulders from reduced muscle perfusion), tremor, and anxiety-like symptoms driven by catecholamine surges rather than psychological pathology. The anxiety misattribution is so common that 69% of POTS patients in a 4,835-person survey received an anxiety diagnosis before POTS was identified.
What causes or triggers hypovolemic POTS?
POTS rarely appears out of nowhere. Most patients can identify a triggering event, and the list of recognized triggers has expanded considerably in the past five years.
Post-viral illness is the most common identified trigger, reported by roughly 41% of patients even before COVID-19. The pandemic made this link undeniable: studies estimate 2-14% of COVID-19 survivors develop POTS, and a 2025 Stanford evaluation found POTS in 28-30% of long-COVID patients referred for autonomic testing. Proposed mechanisms include molecular mimicry (viral proteins resembling autonomic nerve or receptor proteins, provoking autoimmune attack), direct viral injury to autonomic ganglia or small nerve fibers, and persistent immune activation driving ongoing RAAS dysfunction. Epstein-Barr virus, influenza, and other systemic infections are also recognized triggers.
Concussion and traumatic brain injury are the second most common trigger in younger populations. In one series of 268 concussion patients aged 8-25, 7% developed post-concussive orthostatic tachycardia. Over 40% of adolescents evaluated 3 weeks to 6 months after concussion showed evidence of orthostatic tachycardia on testing. Brainstem dysfunction, vagal nerve impairment, and disrupted autonomic regulation are the proposed mechanisms. The overlap between post-concussion syndrome and POTS is substantial — many patients carry both diagnoses, and the symptom profiles (brain fog, fatigue, exercise intolerance, headache) are nearly identical.
Ehlers-Danlos syndrome (hEDS) and other connective tissue disorders create a structural predisposition for POTS. Roughly 31% of POTS patients meet criteria for hEDS, and about 80% of hEDS patients have POTS. The proposed mechanism is that loose, stretchy connective tissue in blood vessel walls allows excessive venous distension and pooling, reducing effective circulating volume. The "triad" of hEDS, POTS, and mast cell activation syndrome (MCAS) is seen frequently in clinical practice.
Mast cell activation syndrome can contribute to functional hypovolemia through histamine-mediated vasodilation and increased capillary permeability (third-spacing of fluid). A 2021 study found MCAS prevalence of 31% among patients with both POTS and hEDS, versus 2% in those without either condition.
Autoimmune mechanisms are under active investigation. Some studies have found elevated autoantibodies targeting adrenergic and muscarinic receptors in POTS patients, which could impair autonomic signaling and RAAS regulation. A 2025 study specifically identified elevated angiotensin II type 1 receptor autoantibodies, which could directly disrupt the volume-retaining arm of the RAAS. However, the clinical significance of these findings remains debated, and a well-controlled 2022 study found no significant autoantibody differences between POTS patients and controls using commercially available assays.
Hormonal factors significantly modify hypovolemic POTS. The menstrual cycle modulates RAAS function, with better orthostatic compensation during the mid-luteal phase when progesterone supports volume retention. About 72% of women with POTS report symptom worsening premenstrually or during menses. Oral contraceptives containing drospirenone (a progestin with anti-mineralocorticoid properties) can worsen hypovolemia and should be specifically avoided.
Deconditioning functions as both a trigger and a perpetuator. Extended bedrest from any cause — surgery, illness, injury, pregnancy — can reduce blood volume enough to unmask POTS in a predisposed individual.
How is hypovolemic POTS diagnosed?
Diagnosis proceeds in two stages: confirming POTS, then identifying the hypovolemic mechanism.
Confirming POTS requires either an active standing test or a head-up tilt table test. The active standing test is simple: measure heart rate and blood pressure supine, then at 1, 3, 5, and 10 minutes of quiet standing. A sustained increase of 30+ bpm (40+ in adolescents) without orthostatic hypotension confirms the diagnosis. The Canadian Cardiovascular Society recommends morning testing when orthostatic tachycardia tends to be most pronounced and advises against testing after large meals, caffeine, or dehydration. Tilt table testing at 60-70 degrees provides a more controlled environment and remains the gold standard in autonomic laboratories.
Identifying hypovolemia is where most clinical evaluations fall short. Several approaches exist:
A 24-hour urine sodium below 100 mmol is the most practical screening test and is the marker used in the 378-patient phenotyping study to classify hypovolemic POTS. It reflects inadequate salt retention by the kidneys and can be ordered by any physician.
Blood volume analysis using the DAXOR BVA-100 is the gold standard. This FDA-cleared test uses iodine-131-labeled albumin injected intravenously, with serial blood sampling over 24 minutes. It separately quantifies total blood volume, plasma volume, and red blood cell volume as deviations from predicted normal values. It is the only direct measure of whether a patient is actually hypovolemic, but availability is limited to specialized centers.
CO-rebreathing is a newer, radiation-free alternative validated at Vanderbilt in 2024 that successfully detected blood volume deficits in POTS patients. It is not yet widely available.
A RAAS panel (plasma renin activity, serum aldosterone) can reveal the characteristic paradox of low aldosterone despite low volume. This is not diagnostic on its own but adds mechanistic clarity.
Additional subtyping tests help identify overlapping mechanisms: supine and standing catecholamines (standing norepinephrine above 600 pg/mL identifies hyperadrenergic POTS), QSART or thermoregulatory sweat test (abnormalities identify neuropathic POTS), and echocardiography or cardiac MRI (identifies the "small heart" of cardiac deconditioning). Comprehensive autonomic testing is ideal but not available everywhere.
The differential diagnosis includes dehydration, anemia, hyperthyroidism, pheochromocytoma, adrenal insufficiency, cardiac arrhythmias, and medication effects. Commonly co-occurring conditions like hEDS, MCAS, and chronic fatigue syndrome are not exclusionary and should be evaluated simultaneously.
A critical context note: the mean diagnostic delay for POTS is 4.9 years, during which patients see an average of 7 physicians. Nearly 60% are told their symptoms are psychosomatic. Women experience significantly longer delays (7.0 vs. 3.8 years), and are 2.7 times more likely to wait over a decade for diagnosis.
What are the treatment options for hypovolemic POTS?
There are no FDA-approved medications for POTS. The 2015 Heart Rhythm Society consensus contains no Level I evidence recommendations, and the total evidence base across all POTS subtypes amounts to roughly 750 patients in 21 randomized trials conducted between 2000 and 2023. Treatment is built on expert consensus, small trials, and clinical experience.
For hypovolemic POTS, treatment targets the volume deficit directly through three pillars: fluid and salt expansion, pharmacological support, and cardiovascular reconditioning.
Volume expansion is the foundation
The first-line intervention is aggressive oral hydration with electrolytes. Guidelines recommend 2-3 liters of fluid daily with 10-12 grams of sodium per day — far more than a standard diet provides. Salt tablets, oral rehydration solutions (Trioral, Normalyte, LMNT, Vitassium), and liberally salting food are all reasonable approaches. A 2021 crossover trial in JACC showed that a high-sodium diet (300 mEq/day) measurably increased total blood volume and plasma volume while reducing standing norepinephrine.
For patients who cannot maintain volume with oral intake alone, intermittent IV saline (typically 1-1.5 liters of normal saline) can provide rapid symptom relief. A study of 57 patients who had failed an average of 3.6 medications showed a 3.1-point reduction in symptom scores with IV saline. However, chronic IV access carries real risks of infection and thrombosis and is not recommended for long-term use.
Pharmacological volume support
Fludrocortisone (0.1-0.2 mg/day) is a synthetic mineralocorticoid that directly addresses the aldosterone deficit by promoting renal sodium and water retention. It is the most commonly prescribed first-line medication for hypovolemic POTS despite having no randomized trial evidence in this population. Side effects include potassium depletion, supine hypertension, ankle swelling, and headache. Potassium monitoring is essential.
Desmopressin (DDAVP) (0.2 mg orally) promotes free water reabsorption through V2 receptors. A randomized crossover trial of 30 patients showed standing heart rate decreased from 109.2 to 101.9 bpm. The primary risk is hyponatremia (low sodium), which limits its use to intermittent dosing with electrolyte monitoring.
Rate control and vasoactive agents
Low-dose propranolol (10-20 mg) is the best-studied POTS medication overall. A crossover trial of 54 patients demonstrated reduced heart rate increment and symptom improvement. The key finding: lower doses work better than higher ones. Higher doses worsen fatigue and exercise intolerance.
Ivabradine is a selective heart rate reducer that works through a different mechanism than beta-blockers (If-channel blockade), slowing the heart without affecting blood pressure, contractility, or exercise capacity. A 2021 double-blind crossover trial of 22 patients showed significant heart rate reduction and quality-of-life improvement. It is particularly attractive for hypovolemic POTS because it avoids the blood pressure lowering and fatigue of beta-blockers.
Midodrine (5-15 mg three times daily) constricts peripheral blood vessels, counteracting venous pooling and increasing venous return. A 2026 meta-analysis of 14 studies (968 patients) reported benefit rates of 46-91%.
Pyridostigmine (30-60 mg three times daily) enhances autonomic nerve signaling and has modest heart rate-lowering effects. GI side effects (cramping, diarrhea) limit its tolerability.
For refractory cases: erythropoietin (10,000 units subcutaneously weekly) can expand red blood cell volume in patients with documented RBC deficits; octreotide (50-200 mcg subcutaneously three times daily) reduces splanchnic blood pooling.
Exercise reconditioning is the single most effective treatment
Structured exercise is the only intervention shown to produce sustained physiological reversal of POTS. The Levine Protocol (also called the Dallas Protocol), developed at UT Southwestern, begins with horizontal exercise only — recumbent cycling, rowing, and swimming — because upright exercise triggers symptom flares in deconditioned patients. Over 3-8 months, the program gradually adds upright exercise as tolerance builds.
The results are striking: in the original trial, 53% of patients who completed the program no longer met POTS diagnostic criteria. Left ventricular mass increased about 12% and blood volume about 7%. In a community-based registry of 251 patients, 71% of completers achieved remission.
The problem is adherence. Dropout rates approach 60%, largely because the early weeks are difficult and patients often feel worse before they feel better. The CHOP Modified Protocol offers slower progression for severely symptomatic and pediatric patients.
Exercise reconditioning works for hypovolemic POTS specifically because it reverses the deconditioning spiral: increased cardiac demand stimulates blood volume expansion, increases heart size and stroke volume, improves autonomic regulation, and reduces the compensatory tachycardia that drives symptoms.
Supporting interventions
Compression garments must extend to the abdomen to be effective. Knee-high stockings alone do not meaningfully improve venous return. Waist-high stockings at 30-40 mmHg or an abdominal binder provide measurable benefit by compressing the large venous reservoirs in the legs and splanchnic circulation.
Head-of-bed elevation (15-30 cm or 4-6 inches) maintains mild orthostatic stress overnight, activating the RAAS and promoting fluid retention — directly addressing the hormonal deficit in hypovolemic POTS.
Dietary strategies include small frequent meals (large meals divert blood to the gut), avoiding alcohol (vasodilator and dehydrating), minimizing refined carbohydrates (can trigger reactive hypoglycemia and worsened pooling), and timing larger sodium intake in the morning when symptoms are typically worst.
Counter-maneuvers like leg crossing, squatting, calf pumping, and muscle tensing while standing can temporarily increase venous return during symptomatic episodes.
Emerging and investigational therapies
Transcutaneous auricular vagus nerve stimulation (tVNS) produced the strongest signal in recent trials. A 2024 sham-controlled randomized trial of 26 patients showed two months of daily tVNS reduced postural heart rate increase to 17.6 bpm versus 31.7 with sham — while also lowering autoantibodies and inflammatory markers.
Immunotherapy (IVIG, subcutaneous immunoglobulin) has shown promise for suspected autoimmune POTS in case series, with one study reporting 50% reduction in symptom scores and 5 of 7 patients returning to work. Randomized trials are underway.
Stellate ganglion block has shown early promise in case reports but remains experimental.
The RECOVER-AUTONOMIC trial — the largest POTS randomized trial ever conducted — is currently testing IVIG, ivabradine, and coordinated nonpharmacologic care in post-COVID POTS patients across multiple centers. Results will reshape the treatment evidence base.
What if the problem is not just volume but the brain's ability to regulate?
Most POTS treatment targets the downstream consequences of autonomic dysfunction: salt to expand volume, medications to slow heart rate, compression to fight pooling. Those tools matter, and the evidence behind them is real. But they share a limitation: none of them address why the autonomic nervous system lost its ability to regulate in the first place.
POTS is not purely a heart problem or a blood pressure problem. It is a nervous system regulation problem. The autonomic nervous system controls heart rate, blood pressure, breathing rhythm, digestion, and the body's response to positional changes. When that system loses its calibration, often after a viral illness, concussion, or neurological injury, the cascade of symptoms follows.
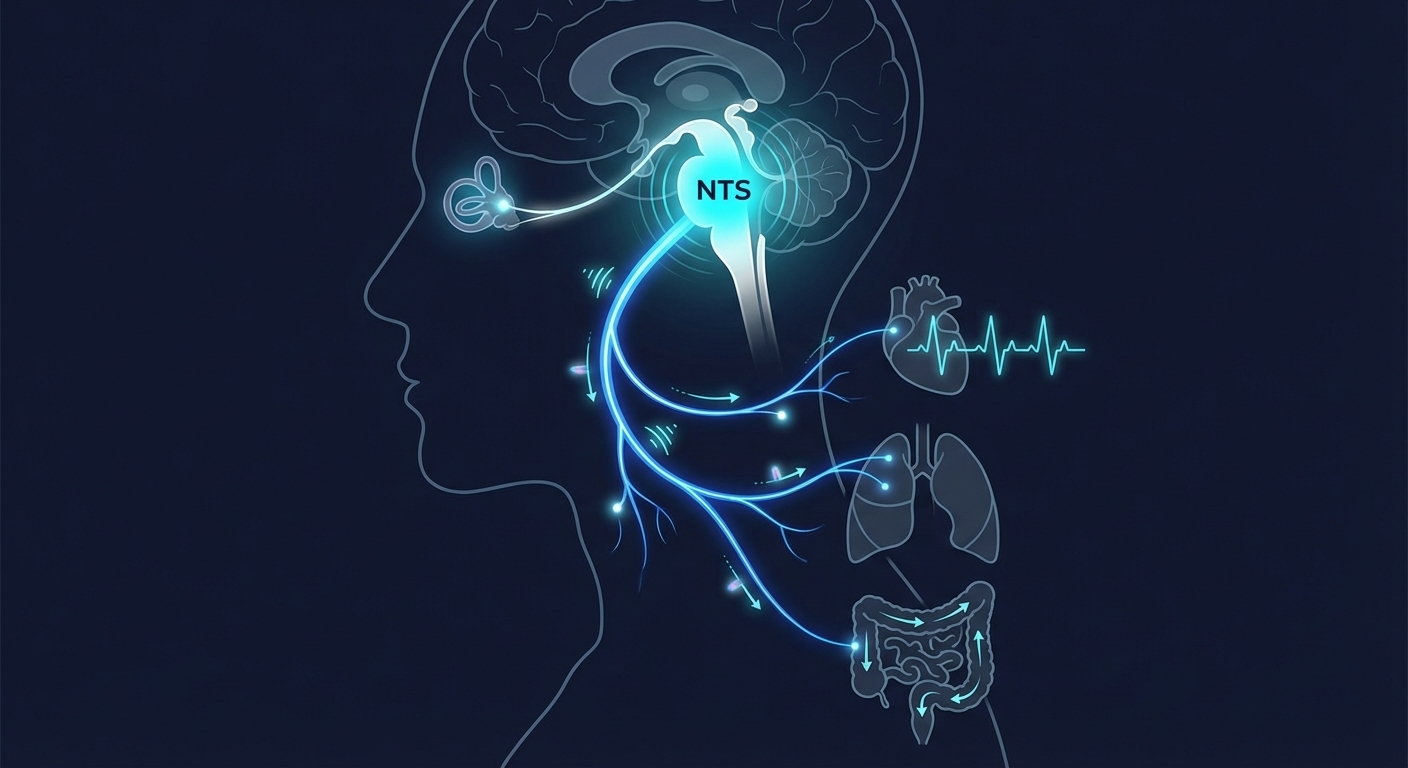
At Cognitive FX, the POTS/ANS Therapy Program approaches dysautonomia through a neurological lens, targeting the central regulatory systems that are dysregulated upstream of the symptoms.
The program runs five days in-clinic. Day 1 begins with a comprehensive evaluation: heart rate, blood pressure, and oxygen saturation (seated and standing), tilt table testing, vestibular assessment, cranial nerve and brainstem (NTS) assessment, supplement screening, and goal setting. This evaluation identifies which POTS subtype or combination of subtypes applies to each patient and maps the specific neurological systems that are misfiring. Treatment begins the same day based on what the evaluation reveals.
Days 2 through 5 are full treatment days, each running approximately six hours of individualized, protocol-driven therapy. The therapies target five key systems that are consistently dysregulated in POTS:
The Nucleus Tractus Solitarius (NTS) is the brainstem's primary integration hub for all autonomic signaling, including cardiovascular, respiratory, and digestive input. In POTS, the NTS is not damaged; it is poorly calibrated. The program uses a structured Cranial Nerve Stack protocol that progressively loads the NTS through its afferent inputs (cranial nerves VII, IX, and X) to improve baroreflex sensitivity and reliability. This is essentially reconditioning applied at the central regulatory level rather than downstream.
The Vestibular System detects gravity, acceleration, and position changes and is in direct communication with the autonomic nervous system. When vestibular processing is inaccurate or delayed, the cardiovascular adjustments needed to maintain brain perfusion on standing misfire. Vestibular training in the program targets both the saccule (vertical motion detection) and utricle (horizontal motion detection) with progressive loading from supine to seated to standing positions.
Breathing Mechanics and CO2 Tolerance are among the most overlooked drivers of autonomic dysfunction. Dysregulated breathing chemistry, particularly low CO2, impairs oxygen delivery to tissues (via the Bohr effect) and perpetuates sympathetic overdrive. The program includes resonance frequency breathing at approximately 6 breaths per minute, which entrains baroreflex oscillations at their natural resonance (~0.1 Hz), the strongest non-invasive amplifier of baroreflex gain in the research literature. CO2 tolerance training progressively recalibrates the brainstem's respiratory drive threshold.
Cranial Nerve Function, particularly the nerves governing breathing, heart rate, swallowing, and gut motility, is directly assessed and targeted with specific activation protocols.
The Intermediolateral (IML) Nucleus, the spinal column of preganglionic sympathetic neurons located T1-L2, governs downstream vascular tone. Thoracic spine mobility and isometric training (wall sits, hand grip exercises) support blood flow and sensory input to this region. A 2023 meta-analysis of 270 trials ranked isometric wall sits as 90.5% effective for blood pressure reduction.
The program also includes NeuroCardio Interval Training (NCT), a protocol designed to put the sympathetic and parasympathetic systems at two extremes, full effort and complete rest, to retrain their coordination. Unlike standard graded exercise protocols that take 3-8 months to show results (and have dropout rates approaching 60%), NCT prioritizes recovery quality over interval quantity. The emphasis on full parasympathetic recovery between intervals directly trains the autonomic flexibility that POTS patients have lost.
What makes this approach different from standard POTS management is the integration. Volume expansion, compression, and dietary strategies are still part of the program. But they operate on a stronger foundation when the brain's regulatory systems are also being recalibrated. Patients leave with a structured daily home routine, including a morning cranial nerve protocol performed before getting out of bed, midday exercise sessions with vestibular warm-ups, evening isometric training, and before-bed progressive muscle relaxation and breathing work, all individualized to their specific POTS profile.
For hypovolemic POTS specifically, this matters because the RAAS dysfunction driving the volume deficit is itself a failure of central regulation. Addressing the brainstem's ability to coordinate the autonomic response does not replace fludrocortisone or salt loading. It targets the upstream problem that makes those interventions necessary in the first place.
Schedule a consultation to find out if you are a candidate for the POTS/ANS Therapy Program.
What is the long-term outlook?
The prognosis of POTS has been traditionally described as favorable, with older estimates suggesting about 50% spontaneous improvement within 1-3 years and a prospective Mayo Clinic study showing 37% no longer met tilt criteria at one year.
A 2024 Journal of the American Heart Association study challenged that narrative. In over 225 pediatric-onset POTS patients followed for an average of 9.6 years, only 0.9% — two patients out of 215 — reported being symptom-free in the prior month. The authors suggested previous high remission rates may reflect heart-rate-based definitions that miss ongoing functional impairment.
The practical reality is somewhere between these poles. Most patients improve substantially with appropriate treatment, especially when the hypovolemic mechanism is identified and targeted early. Factors associated with better outcomes include younger age at onset, identifiable viral trigger, shorter time from symptoms to diagnosis and treatment, and adherence to structured exercise programs. Factors predicting a more difficult course include adult onset, comorbid Ehlers-Danlos syndrome or autoimmune disease, hyperadrenergic subtype, and the single most modifiable factor: prolonged diagnostic delay.
Post-COVID POTS appears to carry a relatively favorable trajectory, with 86-94% showing symptomatic improvement over months.
The critical takeaway for hypovolemic POTS specifically is that the volume deficit is addressable. It responds to salt and fluid loading, fludrocortisone, and structured exercise. The deconditioning spiral is reversible. The patients who do best are those whose hypovolemia is identified rather than overlooked, and who receive comprehensive, mechanism-targeted treatment rather than symptom-by-symptom management.
References
- Raj SR, Biaggioni I, Yamhure PC, et al. Renin-aldosterone paradox and perturbed blood volume regulation underlying postural tachycardia syndrome. Circulation. 2005;111(13):1574-1582. PMID: 15781744
- Fu Q, Vangundy TB, Galbreath MM, et al. Cardiac origins of the postural orthostatic tachycardia syndrome. J Am Coll Cardiol. 2010;55(25):2858-2868. PMID: 20579544
- Sheldon RS, Grubb BP, Olshansky B, et al. 2015 Heart Rhythm Society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm. 2015;12(6):e41-e63. PMID: 25980576
- Vernino S, Bourne KM, Stiles LE, et al. Postural orthostatic tachycardia syndrome (POTS): State of the science and clinical care from a 2019 National Institutes of Health Expert Consensus Meeting - Part 1. Auton Neurosci. 2021;235:102828. PMID: 34144933
- Angeli AM, Salonen BR, Ganesh R, et al. Symptom presentation by phenotype of postural orthostatic tachycardia syndrome. Sci Rep. 2024;14(1):205. PMID: 38168762
- Raj SR, Black BK, Biaggioni I, et al. Propranolol decreases tachycardia and improves symptoms in the postural tachycardia syndrome: less is more. Circulation. 2009;120(9):725-734. PMID: 19687359
- Fu Q, Levine BD. Exercise and non-pharmacological treatment of POTS. Auton Neurosci. 2018;215:20-27. PMID: 30001836
- Wells R, Malik V, Brooks AG, et al. Cerebral blood flow and cognitive performance in postural tachycardia syndrome: insights from sustained cognitive stress test. J Am Heart Assoc. 2020;9(24):e017861. PMID: 33280488
- Shaw BH, Stiles LE, Bourne K, et al. The face of postural tachycardia syndrome - insights from a large cross-sectional online community-based survey. J Intern Med. 2019;286(4):438-448. PMID: 30861229
- Coffin ST, Black BK, Biaggioni I, et al. Desmopressin acutely decreases tachycardia and improves symptoms in the postural tachycardia syndrome. Heart Rhythm. 2012;9(9):1484-1490. PMID: 22561596
- Taub PR, Zadourian A, Lo HC, et al. Randomized trial of ivabradine in patients with hyperadrenergic postural orthostatic tachycardia syndrome. J Am Coll Cardiol. 2021;77(7):861-871. PMID: 33602468
- Ormiston CK, Swiatkiewicz I, Taub PR. Postural orthostatic tachycardia syndrome as a sequela of COVID-19. Heart Rhythm. 2022;19(11):1880-1889. PMID: 35853576
- Wang E, Ganti T, Vaou E, Hohler A. The relationship between mast cell activation syndrome, postural tachycardia syndrome, and Ehlers-Danlos syndrome. Allergy Asthma Proc. 2021;42(3):243-246. PMID: 33980338
- Fedorowski A. Postural orthostatic tachycardia syndrome: clinical presentation, aetiology and management. J Intern Med. 2019;285(4):352-366. PMID: 30372565
- Boris JR, Shadiack EC, McCormick EM, et al. Long-term POTS outcomes survey: diagnosis, therapy, and clinical outcomes. J Am Heart Assoc. 2024;13(14):e033485. PMID: 38958137
- Nwazue VC, Raj SR. Confounders of vasovagal syncope: postural tachycardia syndrome. Cardiol Clin. 2013;31(1):89-100. PMID: 23217690

About the author
Lynn GaufinDr. Lynn Gaufin graduated from the University of Utah and then attended medical school at Cornell University in New York City. After medical school he join the Army and was a surgeon in the military before finishing his Neurological Residency at University of California Los Angeles. Dr. Gaufin specializes in cervical and lumbar spine surgery, brain tumors, brain hemorrhages, and treatment of traumatic brain injuries. Dr. Gaufin is one of the emergency trauma neurosurgeons on call at Utah Valley Hospital. Before he began his practice in Utah he saw a significant amount of traumatic brain injuries during his career in the Army and his residency in Los Angeles. As a surgeon who treats individuals who suffer from mild to severe traumatic brain injuries he recognized a problem in the post operative rehabilitation. Individuals who suffered severe trauma would be admitted into speciality facilities where they would receive months of care. But patients who had a more mild trauma would be released and would largely be on their own when it came to restoring their cognitive function. That problem is what lead Dr. Gaufin to team up with Dr. Fong and Dr. Allen in the creation of Cognitive FX. Cognitive FX was able to take the research that Dr. Fong and Dr. Allen started in their Phd programs and bring it into the clinical environment.










