TMS vs. tDCS for Depression: Which Is Better?
For many people living with major depression, antidepressant medications either don’t work or only get them part of the way to recovery. Symptoms may ease for a while, but then return, or never fully...

While many people are learning about Transcranial Magnetic Stimulation (TMS) for the first time, this brain stimulation method has been helping patients for nearly 40 years. Originally developed as an experimental tool for brain research, TMS is now considered an established FDA-approved treatment for major depressive disorder and other mental health conditions—particularly for patients who haven’t found relief with antidepressant medication or psychotherapy.
This article provides a broad overview of TMS for patients considering the treatment. We cover:
The roots of TMS go back nearly two centuries. In 1831, English physicist Michael Faraday discovered that a magnetic field can induce an electrical current—a principle known as electromagnetic induction. Over 150 years later, in 1985, British engineer Anthony Barker applied this principle to the brain, creating the first TMS device. His coil-based system could stimulate the motor cortex every five seconds and was used mainly as a research tool.
By the mid-1990s, researchers were testing TMS for neurological and psychiatric conditions, especially major depression. Multiple clinical trials and meta-analyses confirmed its safety and effectiveness, laying the groundwork for broader adoption.
A pivotal moment came in 2008, when the U.S. Food and Drug Administration (FDA) approved the use of TMS for treatment-resistant depression. Since then, TMS has gained worldwide recognition and additional approvals for other conditions.
The original TMS protocol, known as repetitive TMS (rTMS), targets the dorsolateral prefrontal cortex (DLPFC)—an area of the brain linked to mood regulation. During treatment, a magnetic coil is placed over the scalp to deliver pulses that modulate brain activity.
The typical rTMS protocol involves:
Schedule: 5 sessions per week for 4–6 weeks (20–30 sessions total)
Session length: About 40 minutes
Pulse pattern: 5–10 seconds of stimulation followed by 30–60 seconds of rest
Timeline for improvement: Some patients improve in 2–4 weeks; others notice benefits only after completing treatment
Over the past two decades, rTMS has become widely accepted, and most U.S. health insurance plans now cover it.
TMS is proven as an effective treatment option for reducing symptoms of depression:
rTMS improves symptoms in ~50% of patients with depression, with over 30% achieving remission.
Accelerated TMS and intermittent theta-burst stimulation (iTBS) offer similar results, with ~50% responding and ~30% reaching remission—often faster.
Newer fMRI-guided accelerated protocols have shown even higher success rates:
Maintenance sessions can help preserve results, sometimes for years.
TMS is non-invasive, well-tolerated, and has a strong safety record, including:
Mild side effects: Scalp discomfort, tingling, muscle twitches, mild headache, lightheadedness
Rare side effects: GI discomfort, insomnia, tinnitus, fainting
Seizure risk: Extremely rare—less than 0.01% per session (under 1 in 10,000)
TMS is FDA-approved for:
Off-label uses include bipolar disorder, Parkinson’s disease, epilepsy, and post-traumatic stress disorder (PTSD).
Since 2008, several new TMS methods have been developed to increase safety and efficacy while addressing key limitations of conventional TMS.
The need to attend sessions 5 days per week makes conventional rTMS treatment challenging to complete amidst work, childcare, and other life commitments. In 2018, the FDA approved intermittent theta-burst stimulation (iTBS), which condenses sessions from 37 minutes to about 3 minutes.
Some clinics offer iTBS on the same treatment schedule as rTMS. Sessions take a fraction of the time, but patients still need to visit the clinic 5 days per week for 4 to 6 weeks. Other clinics offer accelerated protocols that deliver multiple sessions per day, completing a full course of treatment in as little as 5 days.
One key limitation of rTMS lies in how technicians determine where to place the magnetic coil. The most common approach—the “5 cm method”—involves clinicians manually measuring the patient’s head to estimate the correct stimulation site. While simple to perform, it does not account for individual differences in head size, shape, or brain structure. Even a deviation of just a few millimeters from the dorsolateral prefrontal cortex (DLPFC) can significantly reduce treatment effectiveness.
To address this, Stanford researchers developed Stanford Intelligent Accelerated Neuromodulation Therapy (SAINT™), an FDA-approved protocol from 2022 now regarded as the gold standard for treatment-resistant depression.
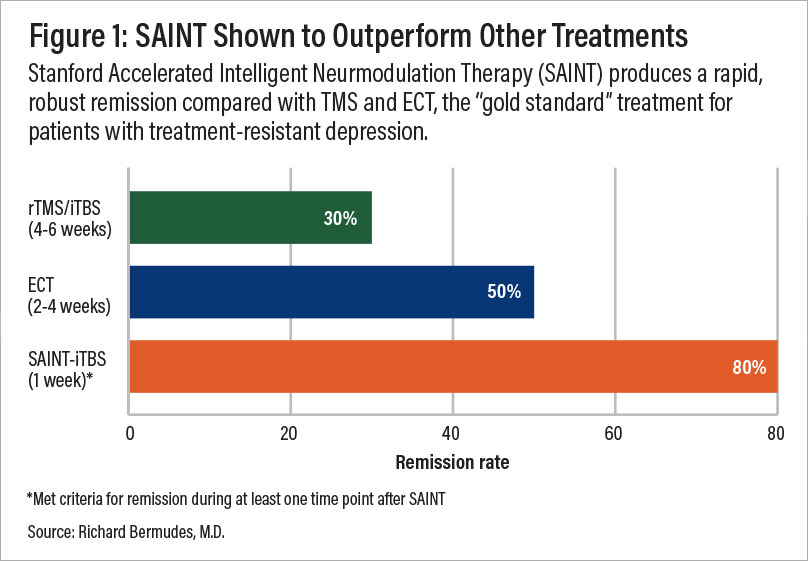
A comparison of remission rates for rTMS/iTBS, electroconvulsive therapy (ECT), and SAINT-iTBS.
SAINT uses functional MRI (fMRI) to pinpoint each patient’s exact DLPFC location based on brain connectivity patterns. During treatment, neuronavigation software ensures the coil is placed over that precise spot for every session, eliminating much of the targeting inconsistency seen with traditional methods.
This precision—combined with an accelerated delivery schedule—has produced remarkable outcomes. The SAINT protocol delivers 10 sessions per day for 5 consecutive days, totaling 50 sessions. Many patients experience rapid and substantial symptom relief.
Our Utah-based clinic, Cognitive FX, offers an accelerated fMRI-guided TMS protocol with the same core elements that make SAINT™ TMS so revolutionary— fMRI and neuronavigated targeting with accelerated iTBS delivery—at a significantly lower cost.
Key differences from Magnus SAINT™ TMS:
We use advanced fMRI analysis by our neuroscientist and physician, rather than proprietary software, to determine the stimulation site.
We process scans in-house, leveraging 25 years of clinical fMRI experience from treating brain injury patients.
We pass those efficiencies on to patients: $9,000–$12,000 vs. $30,000+.
| Accelerated fMRI - TMS | Magnus SAINT™ TMS | |
|---|---|---|
| FDA-Approved iTBS | ✔ | ✔ |
| FDA-Approved Neuronavigators | ✔ | ✔ |
| FDA-Approved Figure 8 Coils | ✔ | ✔ |
| Number of Treatment Days | 5 | 5 |
| Treatments per Day | 10 | 10 |
| Total Treatments | 50 | 50 |
| Number of TMS Pulses | Approx. 90,000 | 90,000 |
| Resting motor threshold pulse intensity | 90–120% | 90–120% |
| FDA-Approved Personalized DLPFC Targeting | ✘ | ✔ |
| Personalized DLPFC Targeting Assists Doctor in Target Location | ✔ | ✘ |
| Personalized E Field Coil orientation | ✔ | ✘ |
| Cost | $9,000 to $12,000 | $30,000+ |
To improve outcomes for our patients, we also include cognitive behavioral therapy (CBT) as part of our treatment. When combined with the traditional TMS (rTMS) method, CBT improved response and remission rates by ~8% and ~19%, respectively. Additionally, CBT is likely to produce sustained improvement over time once treatment has concluded.
Our outpatient, non-invasive treatment is ideal for most patients with major or treatment-resistant depression. For safety, we do not treat:
Click here to learn more about receiving accelerated fMRI TMS therapy at Cognitive FX. Or, take our quiz to see if you’re a good fit for treatment.

Dr. Mark D. Allen holds a Ph.D. in Cognitive Science from Johns Hopkins University and received post-doctoral training in Cognitive Neuroscience and Functional Neuroimaging at the University of Washington. As a co-founder of Cognitive Fx, he played a pivotal role in establishing the unique and exceptional treatment approach. Dr. Allen is renowned for his pioneering work in adapting fMRI for clinical use. His contributions encompass neuroimaging biomarkers development for post-concussion diagnosis and innovative research into the pathophysiology of chronic post-concussion symptoms. He's conducted over 10,000 individualized fMRI patient assessments and crafted a high-intensity interval training program for neuronal and cerebrovascular recovery. Dr. Allen has also co-engineered a machine learning-based neuroanatomical discovery tool and advanced fMRI analysis techniques, ensuring more reliable analysis for concussion patients.

For many people living with major depression, antidepressant medications either don’t work or only get them part of the way to recovery. Symptoms may ease for a while, but then return, or never fully...

Transcranial magnetic stimulation (TMS) and neurofeedback are gaining popularity as non-invasive, medication-free options for treating depression—especially for people who haven’t found relief from ...

If you’re considering brain stimulation therapy for treating major depression, you may wish to understand the differences between repetitive transcranial magnetic stimulation (rTMS) and the more...

Both Transcranial Magnetic Stimulation (TMS) and Vagus Nerve Stimulation (VNS) are FDA-approved neuromodulation treatments for depression. At first glance, they may seem similar—both use electricity...

The type of protocol used is the most important factor influencing the price of TMS. Conventional TMS protocols are often the most affordable, while advanced protocols typically cost more, but can...

If you’re considering Transcranial Magnetic Stimulation (TMS) — a procedure that uses electromagnetic pulses to improve depression symptoms — you may be wondering what the treatment actually feels...